Scientists Develop A Hybrid Way Improve Ca-Ion Energy Storage Devices
Scientists have created a room-temperature Ca-based energy storage devices working on multi-ion reaction mechanism, with the merit of low cost, high capacity, great capability and cycling life as long as 1000 cycles. It potentially promotes the Ca-ion-based devices to next stage.
A research team led by Prof. TANG Yongbing at the Shenzhen Institutes of Advanced Technology (SIAT) of the Chinese Academy of Sciences, in collaboration with Prof. YANG Jinhu from Tongji University, analyzed the dilemma of Ca-ion based technology, and applied a new strategy to solve the difficulty, achieving the best electrochemical performances of Ca-ion energy storage devices so far.
To meet the increasing demand of electric vehicles and intermittent renewable energies, novel energy storage devices with improved performance and affordable cost is critically needed. In comparison to traditional Li-ion batteries, multivalent ion-based devices (Mg2+, Zn2+, Ca2+, Al3+, etc.) are promising options since they may provide high capacity feasibility. Among them, Ca2+ exhibits better kinetics compared to other multivalent chemistries, and therefore denotes a higher power advantage. Taking into account of its abundance merit (2500 times lithium), Ca-ion technology may generate high capacity, high power and low-cost devices.
However, reported calcium-based energy storage systems are still far from satisfactory for practical requirements. Specifically, calcium-based energy storage devices based on the conventional rocking-chair-type mechanism are still meeting many difficulties, i.e., lack of an appropriate combination of suitable electrode materials and electrolytes.
Prof. TANG proposed that multi-ion reaction mechanism is a possible approach to break Ca-technology barrier. Combining the advantage of supercapacitors (good rate and cycling performance) and dual-ion batteries (high capacity and working voltage), a hybrid strategy may lead to devices with improved performance.
Under these consideration, after optimization, a best device, Ca-HSC, is constructed using activated carbon as positive electrode, metal tin foil as negative electrode, and Ca (PF6)2 in carbonate solvent as electrolyte. The capacity retention after 1000 cycles was 84%. That Ca‐HSCs represented the best comprehensive performance of Ca‐ion based energy storage device so far, especially via low‐cost material and simple manufacture technique, which helped the Ca-ion devices one step closer to the next-generation energy storage.
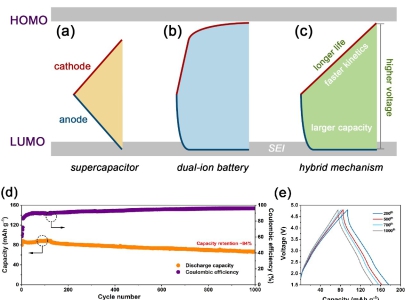
Figure. Three kinds of multi-ion reaction mechanism. Potentials of cathode (red line) and anode (blue line) in a) SC, b) DIB and the c) hybrid mechanism during the charge process, d) Long-term cycling performance of Ca-HSC at current density of 0.2 A g-1, e) Galvanostatic charge-discharge curves of Ca-HSC of 200 to 1000 cycles. (Image by Prof. TANG Yongbing)
Related research is funded by National Natural Science Foundation of China (51822210), Shenzhen Science and Technology Planning Project (Nos. JCYJ20170818153427106, JCYJ20170818153404696, and JCYJ20170818153339619) and published in Advanced Energy Materials (cited as Adv. Energy Mater. 2019, 1803865) .
Link to the paper: https://onlinelibrary.wiley.com/doi/10.1002/aenm.201803865
CONTACT:
ZHANG Xiaomin
Email: xm.zhang@siat.ac.cn
Tel: 86-755-8658529